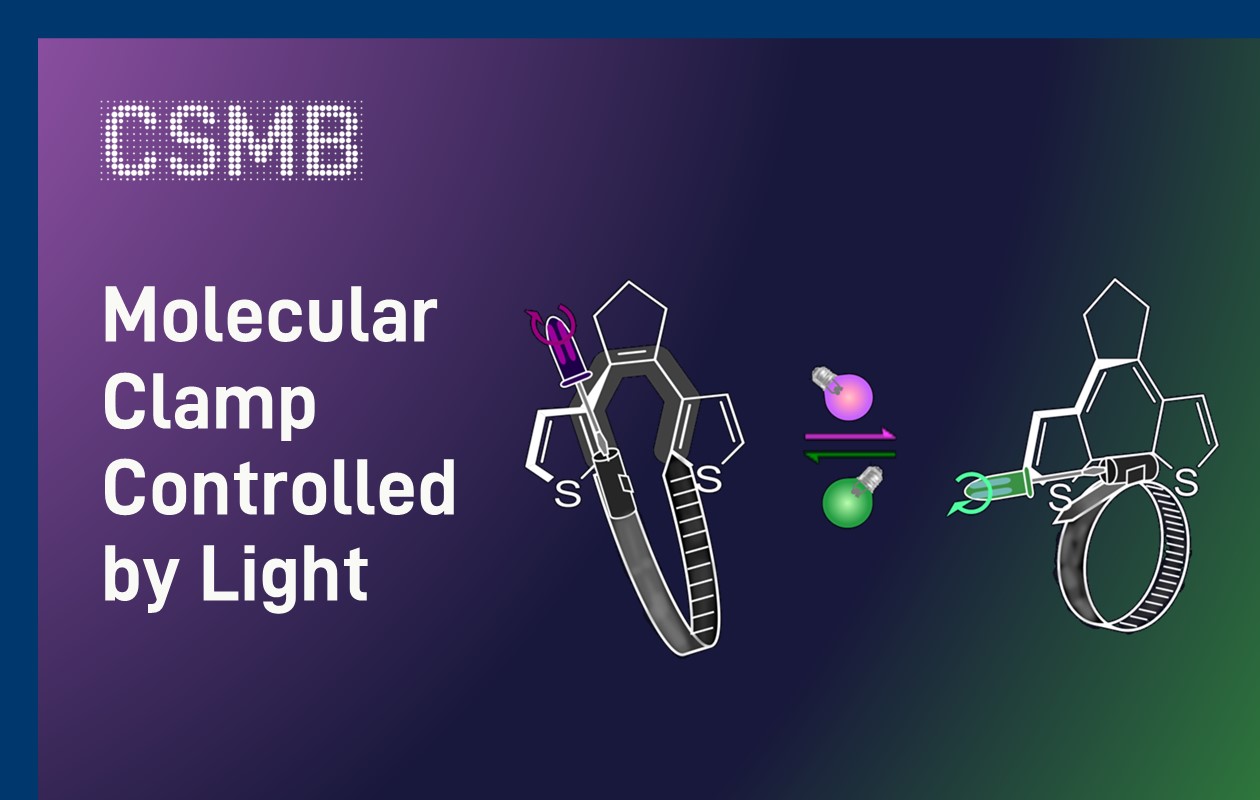
Graphic (molecule and clamps) by First Author Tom Bösking (modified)
Controlling exactly when and where chemical reactions take place remains one of the central challenges in modern chemistry. Heat, mixing, or catalysts typically affect entire systems at once, offering little spatial or temporal precision. A team led by Prof. Stefan Hecht at Humboldt-Universitat zu Berlin (HU) has now developed a strategy that replaces these blunt tools with something far more precise: light. Working in collaboration with partners in Aachen and at the Max Planck Institute, the researchers designed molecules that respond to light by mechanically changing their shape. These so-called photoswitches behave like tiny adjustable clamps. When illuminated, they contract or expand, storing or releasing mechanical strain. This internal tension directly determines how reactive the molecule is - effectively turning chemical reactions on or off.
A long-standing vision
Photoswitchable molecules have been a focus of research at Humboldt-Universitat for decades. The idea is simple but powerful: use light as a clean, fast, and non-invasive external trigger to control molecular function. Yet translating this into precise control over chemical reactivity has remained difficult.
Molecular Clamp
At the center of the study are looped diarylethene photoswitches. Upon irradiation, these molecules undergo a reversible reaction that either closes or opens part of their structure. In doing so, they effectively tighten or loosen a molecular ring, much like adjusting a hose clamp with a screwdriver.
This geometric change alters the internal strain of the molecule. And strain, in turn, governs how readily it undergoes chemical reactions. By switching between two light-induced states, the researchers could modulate reactivity without changing the chemical composition.
Importantly, the team also established a direct way to measure this strain. A shift in the molecule’s optical absorption serves as a diagnostic signal, allowing researchers to "read out" how much tension is stored in the system.
Unexpected Behavior
One surprising finding emerged when comparing the two light-controlled states of the molecule. Intuitively, a more strained molecule might be expected to react faster. However, the experiments showed the opposite trend: the less constrained form reacted significantly faster.
Computational studies helped explain this unexpected result. The open form of the molecule can adopt multiple conformations that lower the barrier for the reaction, while the closed form is structurally more restricted. The work therefore highlights that chemical reactivity in strained systems depends not only on stored energy but also on molecular flexibility and shape.
Toward programmable chemistry
The implications of this work extend well beyond a single reaction type. Because the design relies on simple structural principles, it can be adapted to a wide range of chemical systems.
Materials could be engineered to heal themselves when illuminated at specific locations. In recycling, polymers might be selectively broken down only under defined light conditions. In advanced 3D printing, chemical reactions could be controlled voxel by voxel, enabling unprecedented precision.
In medicine, the concept is particularly compelling. Light could activate drugs only at targeted sites, minimizing side effects while maximizing efficacy. Unlike heat or chemical triggers, light offers unmatched spatial and temporal resolution.
Contact
Prof. Stefan Hecht, Ph.D.
Tel.: +49 (0)30 2093-82650
E-Mail: materialdesignlab.chemie@hu-berlin.de
Article
Bösking T, Schwarz D, Aßenmacher D, Fiukowski O, Pohl M, Pauls M, Bannwarth C, Kolarski D, Hecht S
Light-driven reversible ring contraction and expansion to modulate strain, conformation, and reactivity
Chem, 2026; in print
DOI: 10.1016/j.chempr.2026.102947