Small Molecule Stabilization of Proteins and Nanoparticles: Rethinking Colloidal Stabilizations
Prof. Francesco Stellacci graduated in Materials Engineering at the Politecnico di Milano in 1998 with a thesis on photochromic polymers with Prof. Giuseppe Zerbi and Mariacarla Gallazzi. In 1999 he moved to the Chemistry Department of the University of Arizona for as a post-doc in the group of Joe Perry in close collaboration with the group of Seth Marder. In 2002 he moved to the Department of Materials Science and Engineering at the Massachusetts Institute of Technology as an assistant professor. He was then promoted to associate without (2006) and with tenure (2009). In 2010 he moved to the Institute of Materials at EPFL as a full Professor. He holds the Alcan EP Chair.
Today Stellacci is the head of EPFL’s Supramolecular Nano-Materials and Interfaces Laboratory (SuNMIL) and a fellow of the European Academy of Sciences. He was one of the recipients of the Technology Review TR35 “35 Innovator under 35” award in 2005, and the Popular Science Magazine “Brilliant 10” award in 2007. He has been a Packard Fellow starting 2005. He is a Fellow of the Royal Society of Chemistry, of the Global Young Academy, the European Academy of Sciences, and the Swiss Academy of Engineering.
He received the 2025 Best Teacher Award in the Materials Science and Engineering section for his engaging, hands-on approach that prioritizes real-world experiences over conventional lectures. Stellacci is on sabbatical for the 2025–2026 academic year.
He combines fundamental science research on solid-liquid interfaces with inspiration from Nature to create new molecules and new materials for a variety of applications. SuNMIL’s approach to research is guided by two principles that together constitute its mission:
– Develop novel fundamental science in the field of solid liquid interfaces. In particular, we aim at developing techniques so to advance the knowledge on the fundamental problem of proteins in water.
– Tackle great unresolved challenges for mankind. Currently we are focusing on two problems. The first is the development of broad-spectrum antivirals. The second is an attempt to generate more sustainable polymeric materials.
SuNMIL focuses on problems that plague under-developed or developing countries. For example, millions of people die every year because of viral diseases, most in developing countries. For this reason, we developed a line of research on supramolecular approaches to design novel antivirals. For the same reason, our approach to generating recyclable plastics is rooted on the idea of generating value from waste.
Abstract
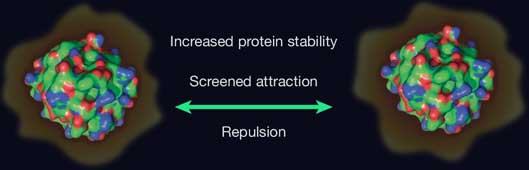 Amino acids have been used for decades in biological and pharmaceutical formulations, yet their stabilizing role is still debated and is often described in non-quantitative or protein-specific terms. In this talk, I will argue that we have been overlooking a general property: amino acids act as modulators of colloidal interactions—not just for proteins, but also for DNA and even synthetic nanoparticles.
Amino acids have been used for decades in biological and pharmaceutical formulations, yet their stabilizing role is still debated and is often described in non-quantitative or protein-specific terms. In this talk, I will argue that we have been overlooking a general property: amino acids act as modulators of colloidal interactions—not just for proteins, but also for DNA and even synthetic nanoparticles.
I will begin by showing how we systematically measured the interactions between suspended particles using osmotic virial coefficients and potential of mean force. We find that the stabilizing effects of amino acids are remarkably consistent across very different systems, suggesting a broad, non-specific mechanism.
To explain this, we developed a theoretical model treating amino acids as weakly interacting small molecules binding to patchy colloidal surfaces. We put this model to the test, comparing predictions against experimental dissociation constants, and the numbers line up: for instance, with proline and lysozyme, theory and experiment agree within a factor of two. [1]
The model leads to some clear, testable predictions—like the idea that short peptides should be more effective than the same number of free amino acids, or that charged amino acids only stabilize oppositely charged proteins. We verified each of these in the lab.
Finally, I will show that these weak interactions can have strong consequences: they can suppress liquid-liquid phase separation, block stress granule formation in cells, and even double insulin’s bioavailability in mice when co-formulated with proline.
Overall, our results suggest we should think of small molecules like amino acids not just as passive ingredients but as active modulators of colloidal behavior—just as we already account for salt when studying electrostatics, we should be paying similar attention to these small molecular players.
Please, let us know if you plan to attend by registering here:
No entry fee.
During the event, photo and video recordings might be made. The material will be used exclusively for the purpose of public relations at CSMB & HU Berlin.